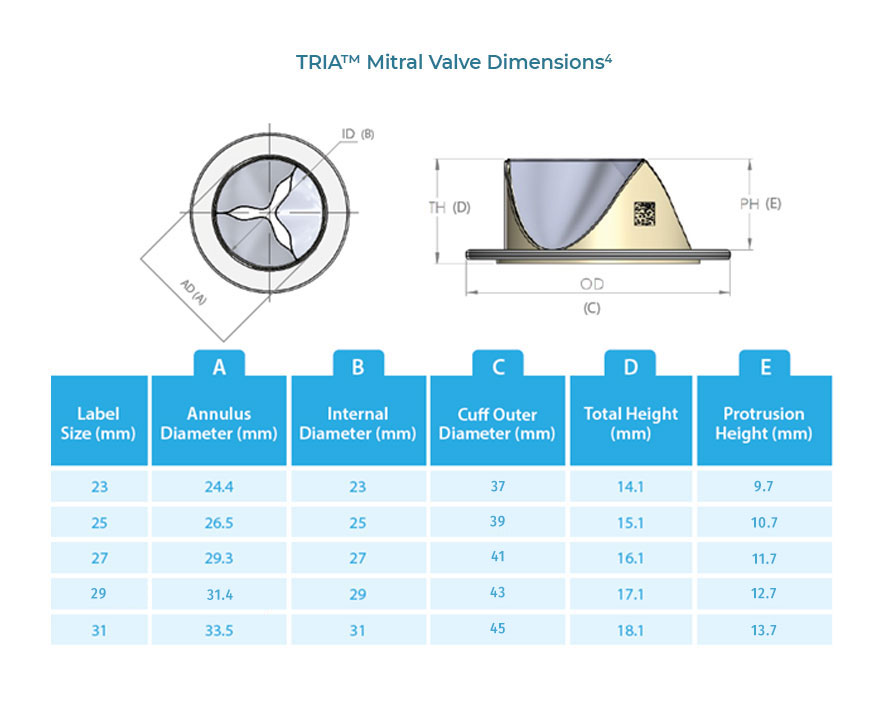
The TRIA™ Mitral Valve is the only replacement heart valve that combines LifePolymer™, computer-aided design, and robotic manufacturing. It is deliberately different—designed to last a lifetime without compromising quality of life.
The mitral valve allows oxygen-rich blood to flow smoothly from the lungs, through the heart, and to the rest of the body. When it stops functioning properly due to stenosis (narrowing) or regurgitation (leakage), it can cause heart failure, arrhythmias, stroke, and other complications. Replacing the valve can restore heart function and enhance quality of life.
Dolphin Life Science India LLP is the legal manufacturer of the TRIA™ Mitral Valve in India. In partnership with Foldax Inc.®, Dolphin brings in-country expertise and advanced manufacturing capability to support the world’s first polymer surgical mitral valve made with LifePolymer™. This collaboration enables localized production that ensures precision, quality, and availability for Indian patients.
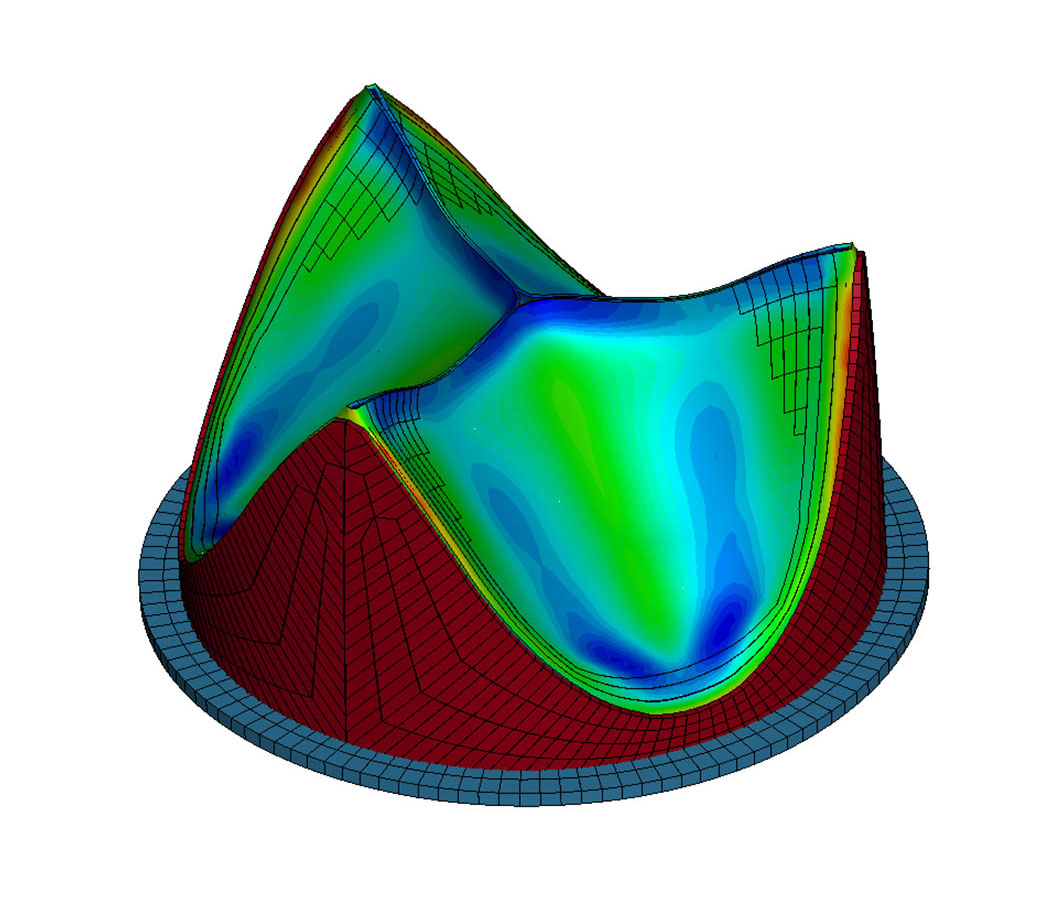
TRIA™ leaflets and frames are computer designed and robotically manufactured to handle the specific blood flow and pressure of the mitral valve.


Strong Performance After 1 Year
A clinical study was conducted with 67 patients in 8 cities across India. The patients were between 19 and 67 years old, and more than 60% were women, including many of childbearing age.
Here’s what was observed one year after surgery with the TRIA™ Mitral Valve: